Pesticide removals in the nitrifying expanded - Bed filter at drinking water treatment plant
The occurrence of pesticides even at low concentrations in drinking water sources might induce potential risks
to public health. This study aimed to investigate the removal mechanisms of eight pesticides by the nitrifying
expanded-bed filter using biological activated carbon media at the pretreatment of a drinking water plant. The
field analysis demonstrated that four pesticides Flutolanil, Buprofezin, Chlorpyrifos, and Fenobucard, were removed at 82%, 55%, 54%, and 52% respectively, while others were not significantly removed. Under controlled
laboratory conditions with continuous and batch experiments, the adsorption onto the biological activated carbon media was demonstrated to be the main removal pathway of the pesticides. The contribution of microorganisms to the pesticide removals was rather limited. The pesticide removals observed in the field reactor was
speculated to be the adsorption on the suspended solids presented in the influent water. The obtained results
highlighted the need to apply a more efficient and cost-effective technology to remove the pesticide in the
drinking water treatment process.

Trang 1

Trang 2

Trang 3

Trang 4

Trang 5
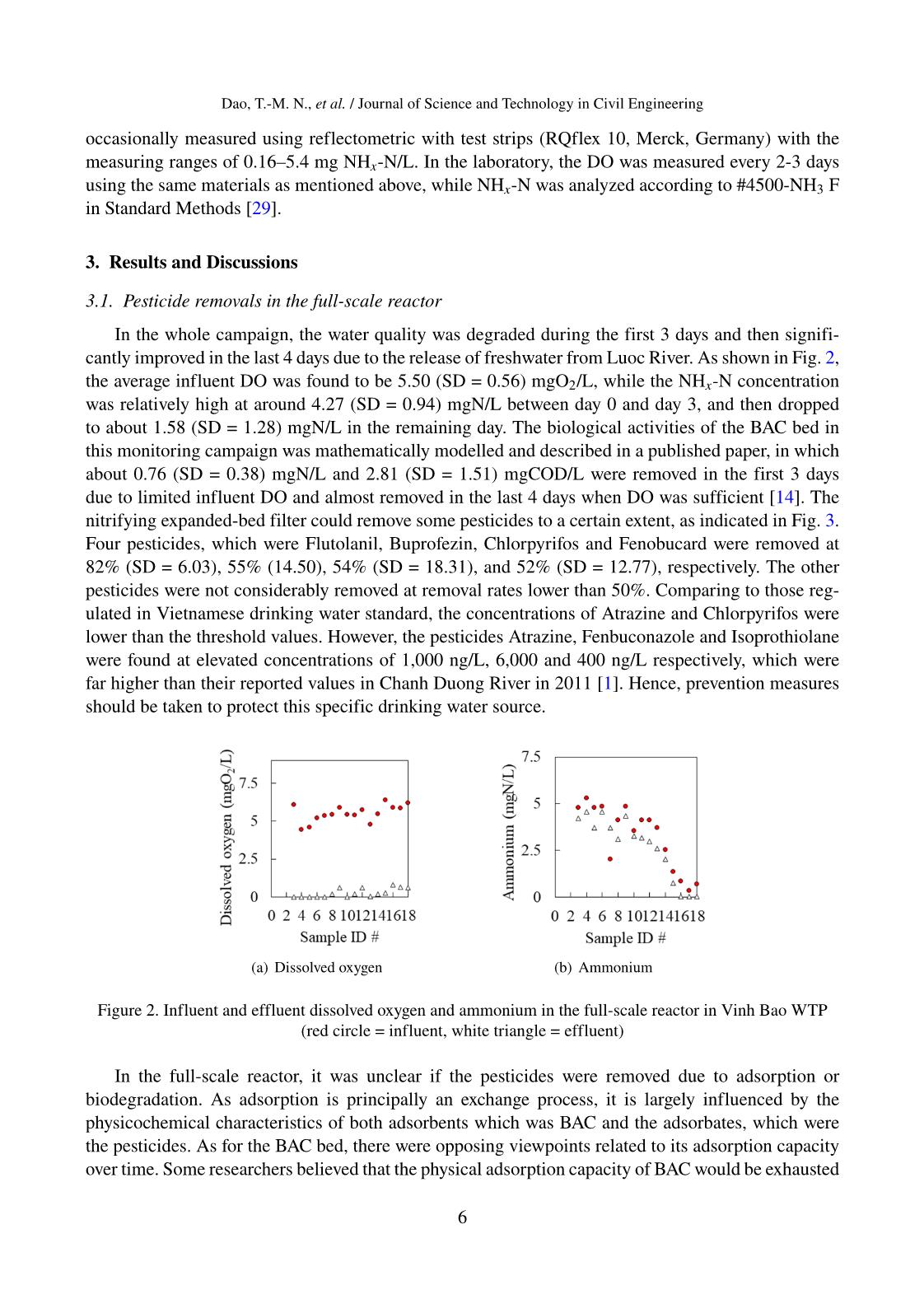
Trang 6
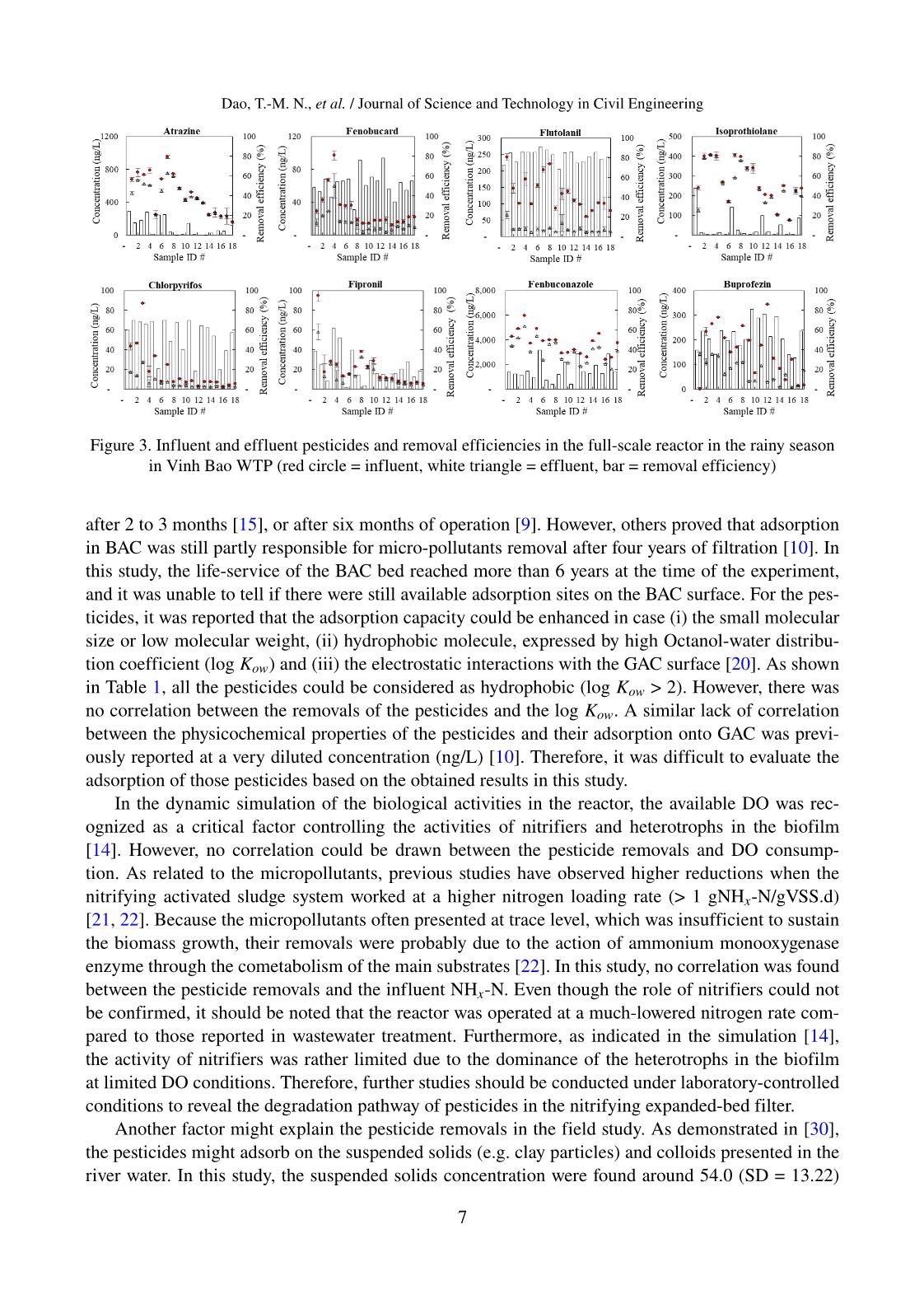
Trang 7
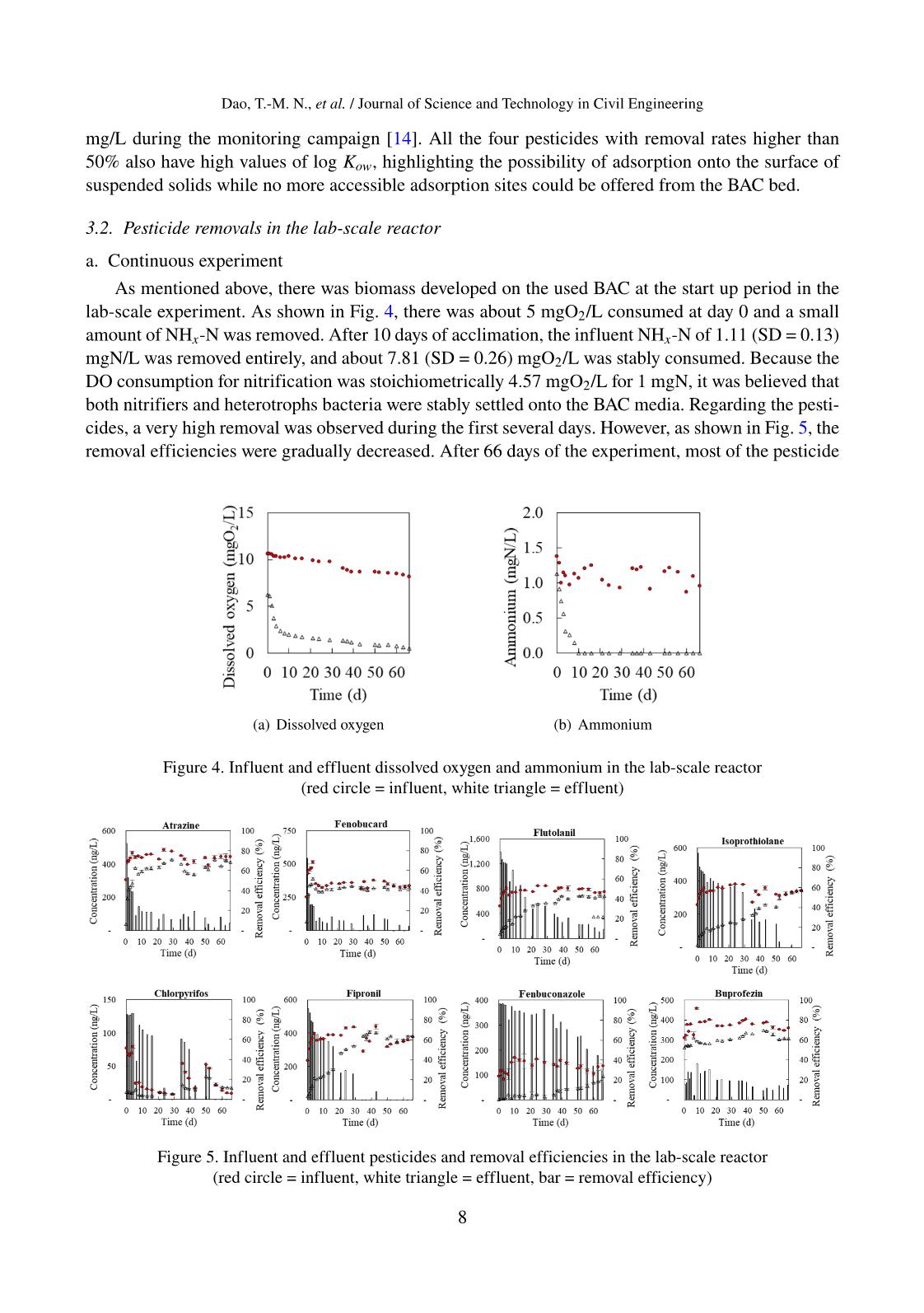
Trang 8
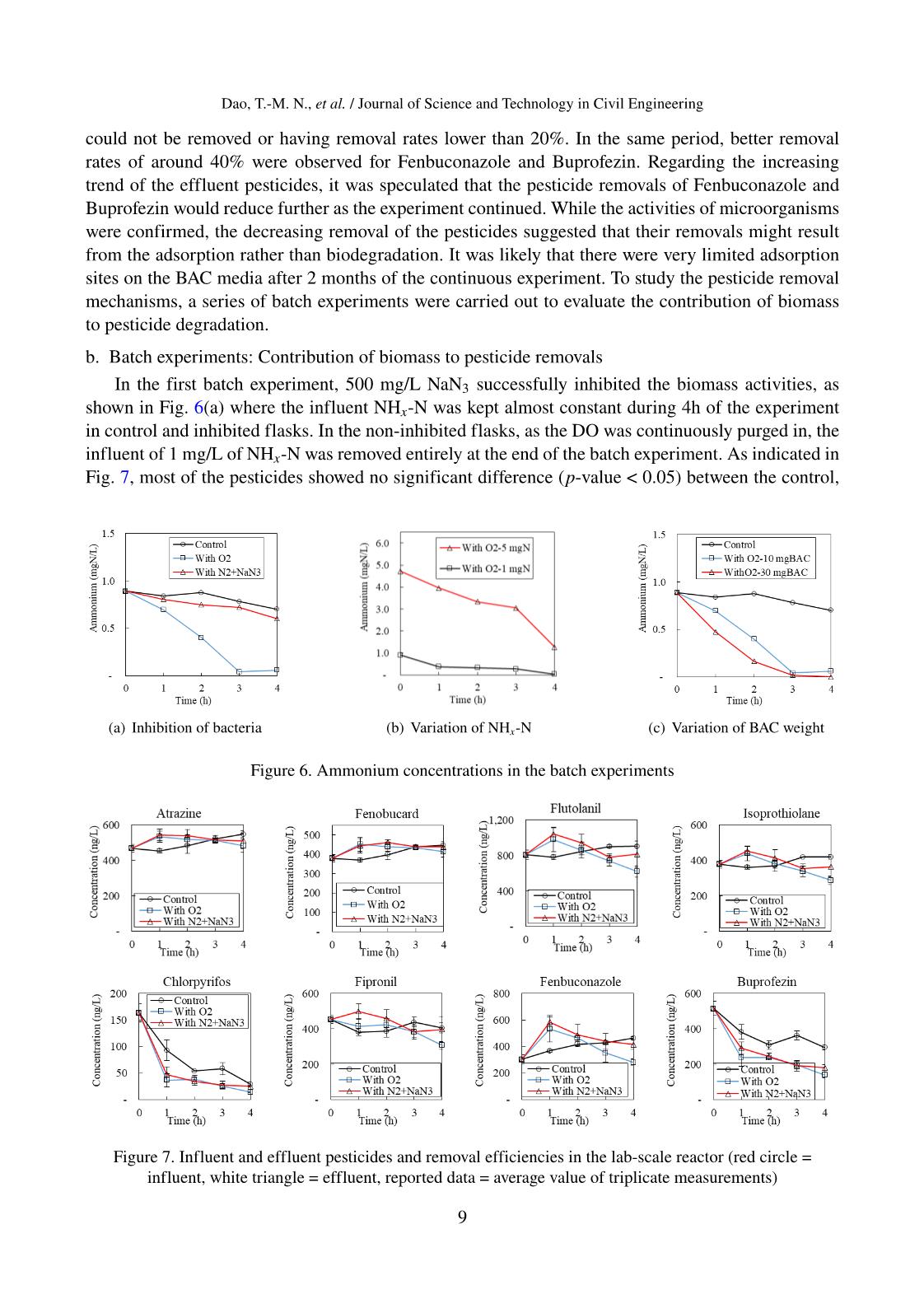
Trang 9
Trang 10
Tải về để xem bản đầy đủ
Tóm tắt nội dung tài liệu: Pesticide removals in the nitrifying expanded - Bed filter at drinking water treatment plant

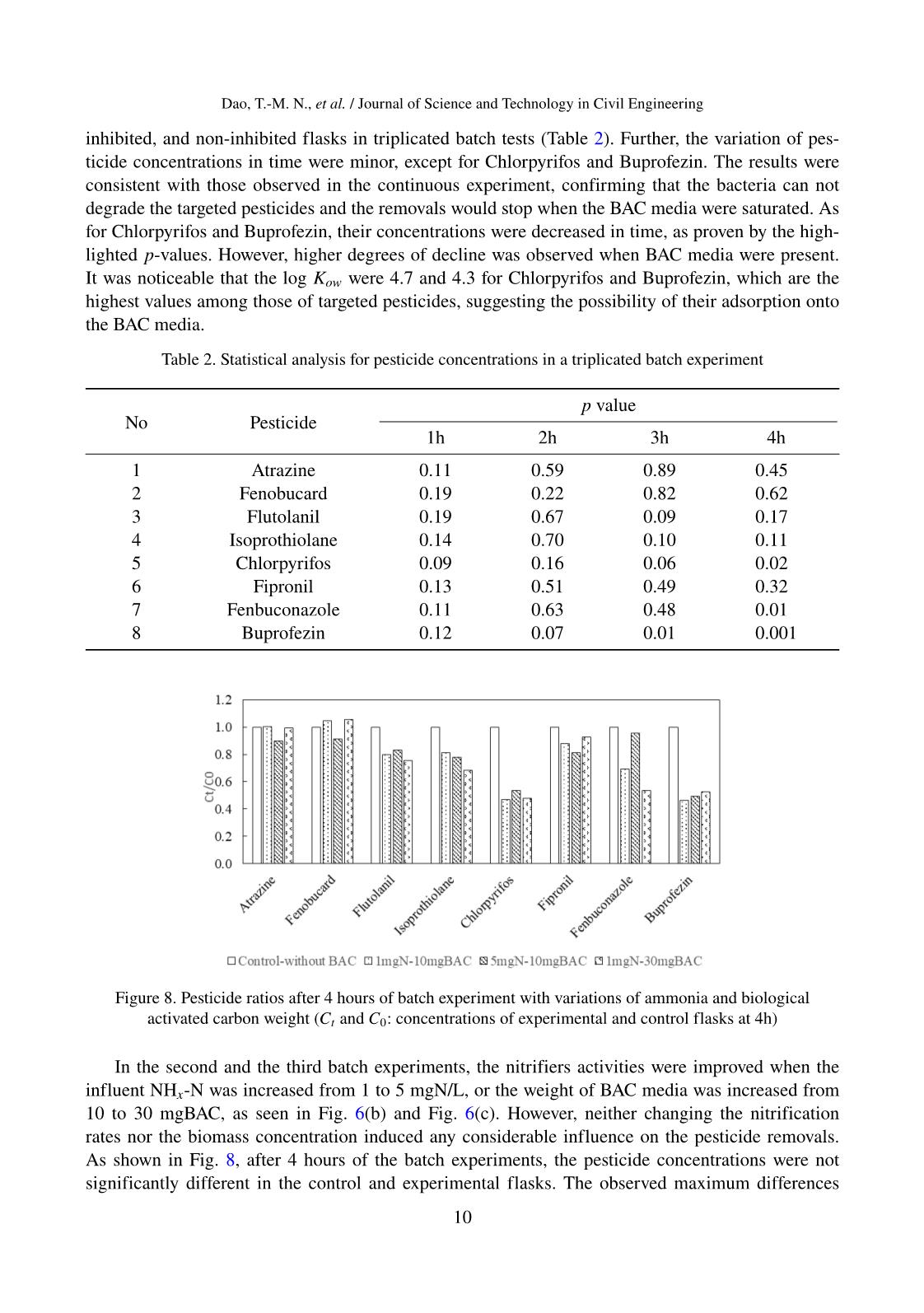
Journal of Science and Technology in Civil Engineering, NUCE 2021 PESTICIDE REMOVALS IN THE NITRIFYING EXPANDED-BED FILTER AT DRINKING WATER TREATMENT PLANT Nguyet Thi-Minh Daoa,b, The-Anh Nguyenc, Viet-Anh Nguyenb, Mitsuharu Terashimaa, Hidenari Yasuia,∗ aFaculty of Environmental Engineering, The University of Kitakyushu, 1-1 Hibikino, Wakamatsu, Kitakyushu City, Fukuoka, Japan bInstitute of Environmental Science and Engineering, National University of Civil Engineering, 55 Giai Phong road, Hai Ba Trung district, Hanoi, Vietnam cFaculty of Water Resources Engineering, Thuy Loi University, 175 Tay Son street, Dong Da district, Hanoi, Vietnam Article history: Received 12/11/2020, Revised 10/12/2020, Accepted 11/12/2020 Abstract The occurrence of pesticides even at low concentrations in drinking water sources might induce potential risks to public health. This study aimed to investigate the removal mechanisms of eight pesticides by the nitrifying expanded-bed filter using biological activated carbon media at the pretreatment of a drinking water plant. The field analysis demonstrated that four pesticides Flutolanil, Buprofezin, Chlorpyrifos, and Fenobucard, were re- moved at 82%, 55%, 54%, and 52% respectively, while others were not significantly removed. Under controlled laboratory conditions with continuous and batch experiments, the adsorption onto the biological activated car- bon media was demonstrated to be the main removal pathway of the pesticides. The contribution of microor- ganisms to the pesticide removals was rather limited. The pesticide removals observed in the field reactor was speculated to be the adsorption on the suspended solids presented in the influent water. The obtained results highlighted the need to apply a more efficient and cost-effective technology to remove the pesticide in the drinking water treatment process. Keywords: biological activated carbon; drinking water treatment; nitrifying expanded-bed filter; pesticide re- moval. © 2021 National University of Civil Engineering 1. Introduction In Vietnam and worldwide, intensive cultivation and increasing application rates of fertilizers, pesticides, herbicides, and other related crop protection products are being practiced to meet the growing food demand and assure food security. Accordingly, the degradations of surface and ground- water, soil, and air quality were observed due to the release of surplus pesticides and herbicides to the environment. The occurrence of pesticides was recorded in the river and groundwater in Hanoi, Hai Phong, Da Nang and Ho Chi Minh Cities [1], as well as in private and public drinking water sources ∗Corresponding author. E-mail address: hidenari-yasui@kitakyu-u.ac.jp (Yasui, H.) 1 Dao, T.-M. N., et al. / Journal of Science and Technology in Civil Engineering in the Mekong Delta in Vietnam [2]; however, their concentrations were lower than the Vietnamese drinking water standard. Scientific evidence has pointed out the public health threats due to the combination of xenobiotic chemicals, even at low concentrations, or the links between organochlorine residues and cancers [3]. Therefore, regulations and guidelines take effect at national and international levels to monitor and control the occurrence and thresholds of emerging chemicals in drinking water. In 2017, the World Health Organization (WHO) had provided the guideline values for 31 pesticides presented in drink- ing water, which were of health significance [4]. In Europe, a proposal for a revised drinking water directive has been adopted since 2018, in which a maximum concentration of 0.1 µg/L for individual pesticides and 0.5 µg/L for total pesticides were regulated [5]. In the United States, the Environ- mental Protection Agency has identified 18 types of pesticides and herbicides with their maximum contaminant levels in the National Primary Drinking Water Regulations [6]. In Vietnam, the Min- istry of Health has recently listed 27 pesticides with their maximum limits in the National Technical Regulation on Drinking Water Quality (QCVN01-1:2018/BTY) [7]. The conventional drinking water treatment process alone, which includes coagulation/flocculation, sedimentation, and filtration, could partly remove the pesticides [8]. Available technologies to remove such persistent chemicals from drinking water sources are chemical oxidation, chemical precipita- tion, membrane, activated carbon adsorption and/or biofilter. Traditionally, granular activated carbon (GAC) was effectively used to adsorb the pesticides and other organic pollutants due to its irregu- lar crevices and porous particle shape that bind those specific contaminants [9]. Thuy et al. (2013) demonstrated that pesticides (Chlorpyrifos, Diazinon, and Carbofuran) could be adsorbed on the low- cost GAC generated from local products (bamboo and coconut shell) at the pilot-scale upgraded from a typical drinking water treatment plant (WTP) ... tch experiments: Contribution of biomass to pesticide removals 323 In the fi st ba ch experiment, 500 mg/L NaN3 successfully inhibited the biomass 324 activities, as shown in Figure 6a where the influent NHx-N was kept almost constant 325 during 4h of the experiment i control and inhibit d flasks. In the non-inhibited flasks, 326 as the DO was continuously purg d in, the influent of 1 mg/L of NHx-N was removed 327 entirely at the end of t e batch experiment. As indicated in Figure 7 , most of the 328 p sticide sh wed no significant diff rence (p-value < 0.05) be ween the control, 329 i hib ted, an non-inhibited flasks in triplicated batch tests (Table 2). Further, the 330 variation of pesticide concentrations in tim w re minor, except for Chl rpyrifos and 331 Buprofezin. The results were consist nt with those observed in the continuous 332 exper ment, confirming th t the bacte ia can not d grad th targete pesticides and the 333 rem vals would stop when the BAC media were saturated. As for Chl rpyrifos and 334 Buprof zin, heir concentrations were decreased in time, as proven by the ighlighted 335 p-values. However, hi h r degre s of d cline as observed when BAC media were 336 present. It was noticeable that th log Kow were 4.7 and 4.3 for Chl rpyrifos and 337 Buprofezin, w ich are the high t values among those of targeted p sticides, su gesting 338 the possibility of their ads rption onto the BAC media. 339 (a) Inhi ition of bacteria (b) Variation of NHx-N (c) Var ation of BAC weight Fig re 6. Ammon um conc ntrations in t e batch experiments 340 Table 2. Statistical analysi for pesticide concentrations in triplicated batch 341 experiment 342 P value No Pesticide 1h 2h 3h 4h 1 Atrazine 0.11 0.59 0.89 0.45 2 Fenobucard 0.19 0.22 0.82 0.62 3 F utolanil 0.19 0.67 0.09 0.17 4 Is prothiolane 0.14 .70 0.10 0.11 5 Chlorpyrifos 0.09 0.16 0.06 0.02 6 Fipronil 0.13 0.51 0.49 0.32 (c) Variation of BAC weight Figure 6. Ammonium concentrations in the batch experiments 16 Figure 7. Influent and effluent pesticides and removal efficiencies in the lab-scale reactor (red circle = influent, white triangle = effluent, reported data = average value of triplicate measurements)Figure 7. Influent and efflue p stici es nd r moval efficiencies in the lab-scale reactor (red circle = influent, white triangle = effluent, reported data = average value of triplicate measurements) 9 Dao, T.-M. N., et al. / Journal of Science and Technology in Civil Engineering inhibited, and non-inhibited flasks in triplicated batch tests (Table 2). Further, the variation of pes- ticide concentrations in time were minor, except for Chlorpyrifos and Buprofezin. The results were consistent with those observed in the continuous experiment, confirming that the bacteria can not degrade the targeted pesticides and the removals would stop when the BAC media were saturated. As for Chlorpyrifos and Buprofezin, their concentrations were decreased in time, as proven by the high- lighted p-values. However, higher degrees of decline was observed when BAC media were present. It was noticeable that the log Kow were 4.7 and 4.3 for Chlorpyrifos and Buprofezin, which are the highest values among those of targeted pesticides, suggesting the possibility of their adsorption onto the BAC media. Table 2. Statistical analysis for pesticide concentrations in a triplicated batch experiment No Pesticide p value 1h 2h 3h 4h 1 Atrazine 0.11 0.59 0.89 0.45 2 Fenobucard 0.19 0.22 0.82 0.62 3 Flutolanil 0.19 0.67 0.09 0.17 4 Isoprothiolane 0.14 0.70 0.10 0.11 5 Chlorpyrifos 0.09 0.16 0.06 0.02 6 Fipronil 0.13 0.51 0.49 0.32 7 Fenbuconazole 0.11 0.63 0.48 0.01 8 Buprofezin 0.12 0.07 0.01 0.001 Journal of Science a d Technology in Civil E gineering, NUCE 2020 p-ISSN 2615- 9058; e-ISSN 2734-9489 17 376 377 378 379 380 Figure 8. Pesticide ratios after 4 hours of batch experiment with variations of 381 ammonia and biological activated carbon weight (Ct and C0: concentrations of 382 experimental and control flasks at 4h) 383 4. Conclusions 384 In this study, the pesticide occurrence and removals were monitored in a full-385 scale nitrifying expanded-bed reactor using BAC media. Although the recorded 386 concentrations might not be a concern for the WTP operators following current 387 Vietnamese regulation, detailed monitoring programs on pesticides would be needed to 388 protect the supply source from these pollutants. The full-scale reactor receiving river 389 water could remove four pesticides, which were Flutolanil, Buprofezin, Chlorpyrifos, 390 and Fenobucard at removal rates of 82%, 55%, 54%, and 52% respectively. 391 While previous studies on wastewater treatment have reported a positive 392 association between nitrification rates and pesticide removals, this research successfully 393 demonstrated that the nitrifying expanded-bed reactor using BAC media was not 394 effective in removing the pesticides in drinking water treatment. In the controlled 395 Figure 8. Pesticide ratios after 4 hours of batch experiment with variations of ammonia and biological activated carbon weight (Ct and C0: concentrations of experimental and control flasks at 4h) In the second and the third batch experiments, the nitrifiers activities were improved when the influent NHx-N was increased from 1 to 5 mgN/L, or the weight of BAC media was increased from 10 to 30 mgBAC, as seen in Fig. 6(b) and Fig. 6(c). However, neither changing the nitrification rates nor the biomass concentration induced any considerable influence on the pesticide removals. As shown in Fig. 8, after 4 hours of the batch experiments, the pesticide concentrations were not significantly different in the control and experimental flasks. The observed maximum differences 10 Dao, T.-M. N., et al. / Journal of Science and Technology in Civil Engineering were around 20% for Flutolanil, Isoprothiolane, and Fenbuconazole. While the role of nitrifiers in nitrifying activated sludge system was confirmed regarding the pesticide degradation [21–24], their insignificant contribution to the pesticide removals observed in this study might due to the different biomass composition in oligotrophic biofilm compared to those in wastewater treatment. Again, only Chlorpyrifos and Buprofezin showed a consistent decrease of concentrations in time. As for the others, the slight variations of concentration might be due to the analytical errors and instrument sensitivity, which were acceptable at these ranges of concentration. From the batch experiments, the pesticide removals in the nitrifying expanded-bed reactor using BAC media was revealed. The adsorption was thought to be the main removal pathway of the pesti- cides. The used BAC media showed good pesticide removal efficiencies at the first period; however, most of the removals were gradually decreased to lower than 20% after 2 months of continuous op- eration. Although the nitrifiers were effective in removing the NHx-N, their contribution to pesticide removals was rather limited. The removals of some pesticides observed in the field reactor might result from the adsorption on the suspended solids presented in the influent water 4. Conclusions In this study, the pesticide occurrence and removals were monitored in a full-scale nitrifying expanded-bed reactor using BACmedia. Although the recorded concentrations might not be a concern for the WTP operators following current Vietnamese regulation, detailed monitoring programs on pesticides would be needed to protect the supply source from these pollutants. The full-scale reactor receiving river water could remove four pesticides, which were Flutolanil, Buprofezin, Chlorpyrifos, and Fenobucard at removal rates of 82%, 55%, 54%, and 52%, respectively. While previous studies on wastewater treatment have reported a positive association between ni- trification rates and pesticide removals, this research successfully demonstrated that the nitrifying expanded-bed reactor using BAC media was not effective in removing the pesticides in drinking wa- ter treatment. In the controlled laboratory conditions, the batch experiments using microbial inhibitors or changing the nitrification rates and biomass concentrations showed insignificant differences in the removals of trace pesticides. Consequently, the adsorption onto BAC media was the main removal pathway for pesticides, which was quickly saturated after 2 months of operation. The obtained re- sults highlighted the need to apply a more efficient and cost-effective technology targeting pesticide removals in drinking water treatment. Acknowledgements This work was supported by JSPS Grants-in-Aid for Scientific Research (18K11739, 19H01160, and 19H02278), Japan. References [1] Hanh, D. T. (2015). Occurrence of organic micro-pollutants in the aquatic environment in Vietnam. Doctoral Thesis, the University of Kitakyushu, Japan. [2] Chau, N. D. G., Sebesvari, Z., Amelung,W., Renaud, F. G. (2015). Pesticide pollution of multiple drinking water sources in the Mekong Delta, Vietnam: evidence from two provinces. Environmental Science and Pollution Research, 22(12):9042–9058. [3] Carvalho, F. P. (2006). Agriculture, pesticides, food security and food safety. Environmental Science & Policy, 9(7-8):685–692. 11 Dao, T.-M. N., et al. / Journal of Science and Technology in Civil Engineering [4] World Health Organization (2017). Guidelines for Drinking-water Quality. [5] European Commission (2018). Climate Change 2013 - The Physical Science Basis. Summary for Policy- makers. In Intergovernmental Panel on Climate Change (Ed.), pp. 1–30. [6] US Environmental Protection Agency (2009). Drinking Water Contaminants. National Primary Drinking Water Regulations. [7] QCVN 01-1:2018/BYT (2019). National technical regulation on Domestic Water Quality. Ministry of Health, Vietnam. [8] Pham, T. T., Nguyen, V. A., der Bruggen, B. V. (2013). Pilot-Scale Evaluation of GAC Adsorption Using Low-Cost, High-Performance Materials for Removal of Pesticides and Organic Matter in Drinking Water Production. Journal of Environmental Engineering, 139(7):958–965. [9] Simpson, D. R. (2008). Biofilm processes in biologically active carbon water purification. Water Re- search, 42(12):2839–2848. [10] Rattier, M., Reungoat, J., Gernjak, W., Joss, A., Keller, J. (2012). Investigating the role of adsorption and biodegradation in the removal of organic micropollutants during biological activated carbon filtration of treated wastewater. Journal of Water Reuse and Desalination, 2(3):127–139. [11] der Bruggen, B. V., Everaert, K., Wilms, D., Vandecasteele, C. (2001). The use of nanofiltration for the removal of pesticides from groundwater: an evaluation. Water Supply, 1(2):99–106. [12] Korotta-Gamage, S. M., Sathasivan, A. (2017). A review: Potential and challenges of biologically acti- vated carbon to remove natural organic matter in drinking water purification process. Chemosphere, 167: 120–138. [13] Alvarino, T., Suarez, S., Lema, J., Omil, F. (2018). Understanding the sorption and biotransformation of organic micropollutants in innovative biological wastewater treatment technologies. Science of the Total Environment, 615:297–306. [14] Dao, N. T.-M., Nguyen, T.-A., Nguyen, V.-A., Terashima, M., Goel, R., Yasui, H. (2020). A mathe- matical model of a nitrifying expanded-bed reactor for the pretreatment of drinking water. Biochemical Engineering Journal, 158:107561. [15] Servais, P., Billen, G., Bouillot, P. (1994). Biological colonization of granular activated carbon filters in drinking-water treatment. Journal of Environmental Engineering, 120(4):888–899. [16] Herzberg, M., Dosoretz, C. G., Tarre, S., Green, M. (2003). Patchy Biofilm Coverage Can Explain the Potential Advantage of BGAC Reactors. Environmental Science & Technology, 37(18):4274–4280. [17] van der Aa, L. T. J., Kolpa, R. J., Rietveld, L. C., van Dijk, J. C. (2012). Improved removal of pesticides in biological granular activated carbon filters by pre-oxidation of natural organic matter. Journal of Water Supply: Research and Technology-Aqua, 61(3):153–163. [18] Liang, C.-H., Chiang, P.-C., Chang, E.-E. (2007). Modeling the behaviors of adsorption and biodegrada- tion in biological activated carbon filters. Water Research, 41(15):3241–3250. [19] Zearley, T. L., Summers, R. S. (2012). Removal of Trace Organic Micropollutants by Drinking Water Biological Filters. Environmental Science & Technology, 46(17):9412–9419. [20] Rattier, M., Reungoat, J., Gernjak, W., Keller, J. (2012). Organic micropollutant removal by biological activated carbon filtration: a review. In Urban Water Security Research Alliance Technical Report. [21] Tran, N. H., Urase, T., Kusakabe, O. (2009). The characteristics of enriched nitrifier culture in the degra- dation of selected pharmaceutically active compounds. Journal of Hazardous Materials, 171(1-3):1051– 1057. [22] Fernandez-Fontaina, E., Omil, F., Lema, J. M., Carballa, M. (2012). Influence of nitrifying conditions on the biodegradation and sorption of emerging micropollutants. Water Research, 46(16):5434–5444. [23] Rattier, M., Reungoat, J., Keller, J., Gernjak, W. (2014). Removal of micropollutants during tertiary wastewater treatment by biofiltration: Role of nitrifiers and removal mechanisms. Water Research, 54: 89–99. [24] Park, J., Yamashita, N., Wu, G., Tanaka, H. (2017). Removal of pharmaceuticals and personal care prod- ucts by ammonia oxidizing bacteria acclimated in a membrane bioreactor: Contributions of cometabolism and endogenous respiration. Science of The Total Environment, 605-606:18–25. [25] Nakamoto, N. (2014). Progress in Slow Sand and Alternative Biofiltration Processes. Water Intelligence 12 Dao, T.-M. N., et al. / Journal of Science and Technology in Civil Engineering Online, 13. [26] Tomlin, C. D. S. (2013). The pesticide manual: A world compendium. Thirteen edition, British Crop Production Council. [27] Team, R. C. (2020). R: A Language and Environment for Statistical Computing. Retrieved from https://www.r-project.org. [28] Duong, H. T., Kadokami, K., Pan, S., Matsuura, N., Nguyen, T. Q. (2014). Screening and analysis of 940 organic micro-pollutants in river sediments in Vietnam using an automated identification and quantifica- tion database system for GC–MS. Chemosphere, 107:462–472. [29] APHA/AWWA/WEF (2017). Standard Methods for the Examination of Water and Wastewater. In Stan- dardMethods (23th ed.). American Public Health Association, AmericanWaterWorks Association,Water Environment Federation. [30] Pham, T. T. (2012). Assessment of Low Cost - High Performance Adsorbents for Safe Drinking Water Production from Polluted Surface Water. Application in Northern Vietnam. Doctoral Thesis, KU Leuven, Belgium. 13
File đính kèm:
 pesticide_removals_in_the_nitrifying_expanded_bed_filter_at.pdf
pesticide_removals_in_the_nitrifying_expanded_bed_filter_at.pdf

